Mobile Immunizations to LTC, Skilled Nursing and Rehabilitation Centers
Covid + Flu Shots
Maricopa - Pima - Yavapai - Pinal
About us
Hope Capital LLC dba 911OccMed is a State Contracted Vendor with Arizona Department of Health to provide NO-COST mobile immunizations for the counties of Maricopa, Pima, Pinal and Yavapai.


Services

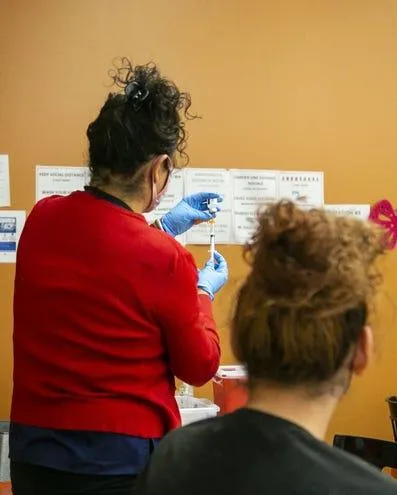
In-Home Mobile Immunizations
Our mobile clinics are designed to meet the specific needs of LTC and skilled nursing facilities. We understand the unique challenges these settings face and provide customized vaccination programs to ensure maximum coverage and protection.
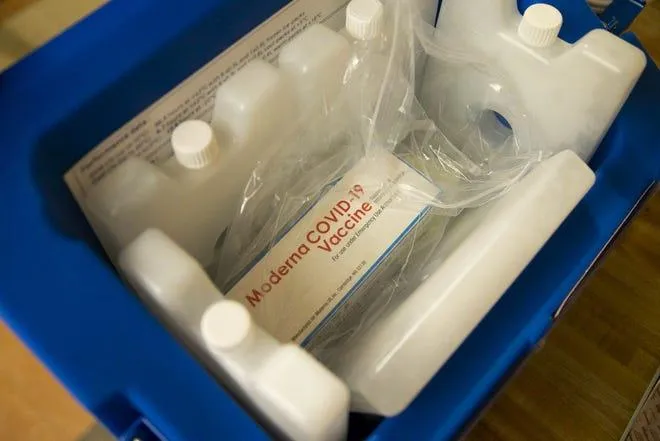
COVID-19 Booster Shots:
Staying Ahead of the Virus: With the evolving nature of the COVID-19 virus, we offer the latest booster shots to ensure continuous protection for your residents and staff against emerging strains.


Flu Vaccinations:
Essential Seasonal Protection: We recognize the critical importance of flu vaccines in safeguarding the health of vulnerable populations in care facilities. Our team ensures timely and effective flu vaccination, perfectly aligned with the flu season.
Expert Medical Team:
Skilled Professionals: Our medical professionals are highly trained and experienced in delivering vaccines safely and efficiently. We prioritize gentle care and informed consent, making the immunization process comfortable and reassuring for all.

Schedule a Call
Our Team

Convenience and Efficiency:
On-Site Services: We bring our services to your doorstep, eliminating the need for transportation and simplifying logistics. Our on-site presence means minimal disruption to your facility’s daily routines.
Comprehensive Coordination:
Hassle-Free Scheduling: From planning to execution, our team works closely with facility administrators to ensure a smooth, well-coordinated vaccination process.
FAQS
About 911OccMed’s Mobile Immunization Services
Q: What types of immunization services does 911OccMed provide?
A: We specialize in administering COVID-19 booster shots and flu vaccinations directly at long-term care (LTC) and skilled nursing facilities. Our mobile clinics are equipped to handle these essential immunizations effectively and safely.
Q: Which areas do you serve?
A: Currently, we serve a variety of regions. Please contact us directly or visit our Service Area page on our website for the most up-to-date information on our coverage areas.
Q: How do you ensure the safety and effectiveness of the vaccines?
A: Our vaccines are sourced from reputable suppliers and are stored and handled according to strict medical guidelines to ensure their safety and efficacy. Our medical staff are trained in the latest immunization practices.
Q: Can 911OccMed handle large-scale vaccination requirements?
A: Absolutely! Our team is well-equipped and experienced in managing large-scale immunization needs, ensuring that both small and large facilities can receive timely and efficient service.
Q: How can our facility schedule your mobile immunization service?
A: Scheduling our services is simple. You can either call us directly or fill out a contact form on our website. Our coordinator will then get in touch with you to discuss your needs and schedule a visit.
Q: What measures does 911OccMed take to manage COVID-19 exposure and safety?
A: We strictly adhere to all current CDC guidelines for COVID-19 to protect our staff and your residents. This includes the use of personal protective equipment (PPE), regular sanitization of equipment, and social distancing practices as feasible.
Q: Is consent required from each resident before vaccination?
A: Yes, informed consent is vital. For residents unable to provide consent themselves, we work with facility administrators to obtain consent from the responsible party or legal guardian.
Q: How are adverse reactions to vaccines handled?
A: Our medical professionals are trained to recognize and manage immediate adverse reactions. We also provide detailed follow-up care instructions and encourage reporting any post-vaccination concerns immediately.
Q: Are your services covered by insurance?
A: Many insurance plans cover COVID-19 and flu vaccines. We can work with you to provide necessary documentation for insurance claims. For specific coverage details, please consult the insurance provider.
Q: What makes 911OccMed different from other immunization service providers?
A: Our commitment to convenience, safety, and efficiency sets us apart. We provide tailored services right at your facility’s doorstep, managed by a team of caring and professional medical experts. We understand the unique needs of LTC and skilled nursing facilities, ensuring a seamless and reassuring experience for everyone involved.
Get In Touch
I consent to receive emails and text messages from 911OccMed regarding information and scheduling for Mobile Immunization Services. Standard message and data rates may apply. For assistance, reply with HELP; to unsubscribe, reply with STOP. Message frequency may vary.
